Answer : The number of moles of gas present in container are 1.697 mole.
Explanation :
Using ideal gas equation,
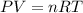
where,
P = pressure of gas = 74 mmHg = 0.097 atm
conversion used : (1 atm = 760 mmHg)
V = volume of gas = 500.0 L
T = temperature of gas =
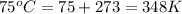
n = number of moles of gas = ?
R = gas constant = 0.0821 L.atm/mole.K
Now put all the given values in the ideal gas equation, we get the number of moles of gas in the container.
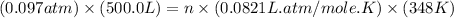
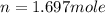
Therefore, the number of moles of gas present in container are 1.697 mole.