Answer:
700 mL or 0.0007 m³
Step-by-step explanation:
P₁ = Initial pressure = 2 atm
V₁ = Initial volume = 350 mL
P₂ = Final pressure = 1 atm
V₂ = Final volume
Here the temperature remains constant. So, Boyle's law can be applied here.
P₁V₁ = P₂V₂
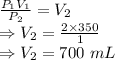
So, volume of this sample of gas at standard atmospheric pressure would be 700 mL or 0.0007 m³