Answer:
The concentration of sodium chloride in an aqueous solution that is 2.23 M and that has a density of 1.01 g/mL is 12.90% by mass
Step-by-step explanation:
2.23 M aqueous solution of NaCl means there are 2.23 moles of NaCl in 1000 mL of solution.
We know that density is equal to ratio of mass to volume.
Here density of solution is 1.01 g/mL.
So mass of 1000 mL solution = (
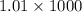 ) g = 1010 g
) g = 1010 g
molar mass of NaCl = 58.44 g/mol
So mass of 2.23 moles of NaCl = (
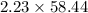 ) g = 130.3 g
) g = 130.3 g
% by mass is ratio of mass of solute to mass of solution and then multiplied by 100.
Here solute is NaCl.
So % by mass of 2.23 M aqueous solution of NaCl =
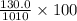 % = 12.90%
% = 12.90%