Answer: The mass of magnesium oxide is greater than the mass of original piece of magnesium.
Step-by-step explanation:
When magnesium burns in air to form a white flaky ash of magnesium oxide, it is undergoing a combination reaction.
A combination reaction is defined as the chemical reaction in which two lighter substances combine to form a single larger substance.
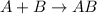
The chemical equation for the formation of magnesium oxide follows:
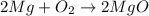
Hence, the mass of magnesium oxide is greater than the mass of original piece of magnesium.