Step-by-step explanation:
Antoine equation is as follows.
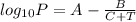
It water is at
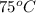 , then values of constants A, B, and C will be as follows.
, then values of constants A, B, and C will be as follows.
A = 8.07131, B = 1730.63, C = 233.426
When pressure, P = 2.5 atm then putting these given values into the above equation as follows.
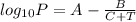
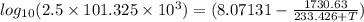
5.40365 =
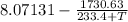
T =
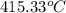
Converting the temperature into Fahrenheit as follows.
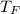 =
=
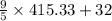
=
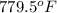
Thus, we can conclude that the boiling temperature of the water is
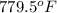 .
.