Answer:
kinetic energy is 3.68 ×
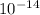 J
J
speed of these electrons is 0.5565 c
Step-by-step explanation:
Given data
voltage difference = 230 kV
to find out
kinetic energy and speed of these electrons
solution
we know kinetic energy formula that is
kinetic energy = qv
kinetic energy = 1.6 ×
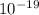 × 230 ×
× 230 ×
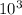
kinetic energy = 3.68 ×
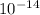 J
J
and
speed of electron that is
kinetic energy = m1c² - m2 c²
here m1 = m / √(1-v/c)²
so
KE/mc² = ( 1 / √(1-(v/c)² ) - 1
put value here
3.68 ×
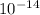 / (3× 10^8)²×9.11×
/ (3× 10^8)²×9.11×
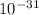 = ( 1 / √(1-v/c)² ) - 1
= ( 1 / √(1-v/c)² ) - 1
0.448835223 = ( 1 / √(1-(v/c)² ) - 1
( 1 / √(1-(v/c)² ) = 0.448835223 + 1 = 1.448835223
√(1-(v/c)² = 1 / 1.448835223 = 0.6902096
(v/c)² = 1 - 0.6902096 = 0.309790
v = 0.5565 c
speed of these electrons is 0.5565 c