Answer: (a) cesium and bromine :
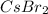
(b) sulfur and barium:
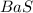
(c) calcium and fluorine :
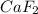
Step-by-step explanation:
For formation of a neutral ionic compound, the charges on cation and anion must be balanced. The cation is formed by loss of electrons by metals and anions are formed by gain of electrons by non metals.
The nomenclature of ionic compounds is given by:
1. Positive is written first.
2. The negative ion is written next and a suffix is added at the end of the negative ion. The suffix written is '-ide'.
(a) cesium and bromine: Here cesium is having an oxidation state of +1 called as
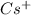 cation and bromine
cation and bromine
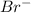 is an anion with oxidation state of -1. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
is an anion with oxidation state of -1. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
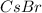 called as cesium bromide.
called as cesium bromide.
(b) sulfur and barium : Here barium is having an oxidation state of +2 called as
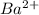 cation and sulphur
cation and sulphur
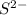 is an anion with oxidation state of -2. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
is an anion with oxidation state of -2. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
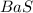 . called as barium sulfide.
. called as barium sulfide.
(c) calcium and fluorine: Here calcium is having an oxidation state of +2 called as
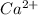 cation and fluorine
cation and fluorine
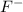 is an anion with oxidation state of -1. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
is an anion with oxidation state of -1. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
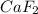 . called as calcium fluoride.
. called as calcium fluoride.