Answer:
final temperature is 300K
Step-by-step explanation:
Given data
temperature TH0 = 450 K
temperature TC0 = 200 K
to find out
final temperature
solution
we consider TH id temperature of hot and TC is temperature of cold object
so
dQH = -mc dTH ..........1
dQC = mc dTC ............2
and for max work
dQH/ TH = dQC/TC
put 1 and 2 here
-mc dTH / TH = mc dTC/ TC
now integrate both side from 450 to T and 250 to T
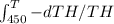 =
=
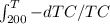
we get ln 450/T = ln T/200
so Temp = √(450*200)
Temp = 300K
final temperature is 300K