Answer:
5.46% is the percent by mass concentration of the solution.
Step-by-step explanation:
Mass of LiBr= 8.2 g(Solute)
Mass of solution = 150 g (Solution)
Mass of solution = Mass of solute + Mass of solvent
Mass of solute < Mass of solvent (Always)
Mass of water = 150 g - 8.2 g = 141.8 g (solvent)
Mass percentage is given as:
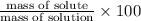
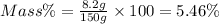
5.46% is the percent by mass concentration of the solution.