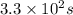Answer: The half-life of a first-order reaction is,
Step-by-step explanation:
All the radioactive reactions follows first order kinetics.
Rate law expression for first order kinetics is given by the equation:
![k=(2.303)/(t)\log([A_o])/([A])](https://img.qammunity.org/2020/formulas/chemistry/college/67w3lufh8bppkbbcp1xaut7f9029vt6k0l.png)
where,
k = rate constant = ?
t = time taken = 440 s
![[A_o]](https://img.qammunity.org/2020/formulas/chemistry/high-school/38eb24kf04xqy5t88y9g0vzh3m04r4nqgg.png) = initial amount of the reactant = 0.50 M
= initial amount of the reactant = 0.50 M
[A] = left amount = 0.20 M
Putting values in above equation, we get:
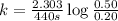
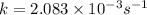
The equation used to calculate half life for first order kinetics:
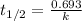
Putting values in this equation, we get:
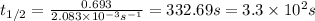
Therefore, the half-life of a first-order reaction is,