Answer:
The volume will not contract to one fourth of the original.
Step-by-step explanation:
Applying Charles's Law as:
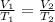
Given ,
V₁ = ?
V₂ = 2 L
T₁ = 5 °C
T₂ = 20 °C
The conversion of T( °C) to T(K) is shown below:
T(K) = T( °C) + 273.15
So,
T₁ = (5 + 273.15) K = 278.15 K
T₂ = (20 + 273.15) K = 293.15 K
Using Charles law as:
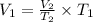
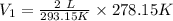
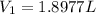
Thus, the volume will not contract to one fourth of the original. (1/4 of 2 L is 0.5 L).