Answer:
a) 90.40 kPa
b) ΔV=0.0220
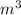
c) ΔH=17.42 kJ
Step-by-step explanation:
As R-134a is a simple fluid, we just need 2 thermodynamic properties to know its state. For the initial state, we know the temperature, and we can calculate the pressure.
Pressure calculation:
If the piston-cylinder system is under atmospheric pressure, the pressure inside the the system may be the sum of the atmospheric pressure plus the pressure that is made by the piston weight:
Piston weight:
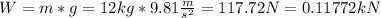
This weight is supported by the fluid through an area of:
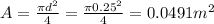
So the pressures that the piston exert over the fluid is:
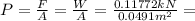
So, the total pressure will be:
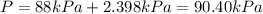
Volume change
The pressure calculated before is constant because the system is not rigid and the atmospheric pressure and the piston pressure does not change. So, knowing temperature and pressure, it is possible to know the specific volume of the fluid by means of the steam tables.
The initial volume is the volume estimated at T=-10ºC and P=90.40 kPa. We realize that it is a superheated vapor because the pressure is a higher than the saturation one. We will have to make an interpolation for several data.
The interpolation for the volume is:
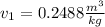
For the final volume (T=15ºC, P=90.40kPa) it will be required a double interpolation, which result is:
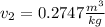
The change in the volume will be:
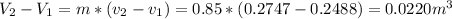
Enthalpy Change
For the enthalpy change we apply the same process, being the interpolation results:
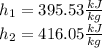
So, the total enthalpy change is:
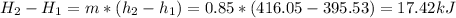
Note: The steam tables used was the tables from van Wylen, 6 edition, table B.5.2