Answer : The values of
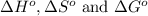 are
are
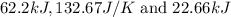 respectively.
respectively.
Explanation :
The given balanced chemical reaction is,
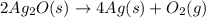
First we have to calculate the enthalpy of reaction
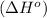 .
.
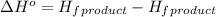
![\Delta H^o=[n_(Ag)* \Delta H_f^0_((Ag))+n_(O_2)* \Delta H_f^0_((O_2))]-[n_(Ag_2O)* \Delta H_f^0_((Ag_2O))]](https://img.qammunity.org/2020/formulas/physics/college/jwg53x8mpln7874vi197xf7damdc4901hf.png)
where,
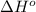 = enthalpy of reaction = ?
= enthalpy of reaction = ?
n = number of moles
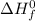 = standard enthalpy of formation
= standard enthalpy of formation
Now put all the given values in this expression, we get:
![\Delta H^o=[4mole* (0kJ/mol)+1mole* (0kJ/mol)}]-[2mole* (-31.1kJ/mol)]](https://img.qammunity.org/2020/formulas/physics/college/q3x5qa1nx9vvz0n74850d3xw0q8ak925xa.png)
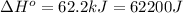
conversion used : (1 kJ = 1000 J)
Now we have to calculate the entropy of reaction
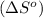 .
.
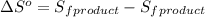
![\Delta S^o=[n_(Ag)* \Delta S_f^0_((Ag))+n_(O_2)* \Delta S_f^0_((O_2))]-[n_(Ag_2O)* \Delta S_f^0_((Ag_2O))]](https://img.qammunity.org/2020/formulas/physics/college/8hfa2v8s4rvdsw1kugxmwy1prvr0xzyuxt.png)
where,
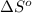 = entropy of reaction = ?
= entropy of reaction = ?
n = number of moles
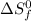 = standard entropy of formation
= standard entropy of formation
Now put all the given values in this expression, we get:
![\Delta S^o=[4mole* (42.55J/K.mole)+1mole* (205.07J/K.mole)}]-[2mole* (121.3J/K.mole)]](https://img.qammunity.org/2020/formulas/physics/college/sviewyzfou37hlmszp7mgwl39jmskcp5u7.png)
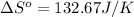
Now we have to calculate the Gibbs free energy of reaction
 .
.
As we know that,
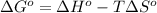
At room temperature, the temperature is 298 K.
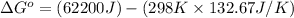
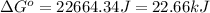
Therefore, the values of
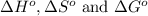 are
are
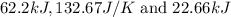 respectively.
respectively.