Answer : The mass of water heated will be, (C) 21.5 kg
Explanation : Given,
Initial temperature =
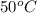
Final temperature =

The value of latent heat of vaporization of steam at
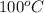 = 2260 kJ/kg
= 2260 kJ/kg
Specific heat of water =
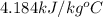
Formula used :
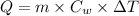
or,
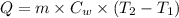
where,
Q = latent heat of vaporization of steam
m = mass of water
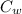 = specific heat of water
= specific heat of water
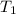 = initial temperature
= initial temperature
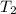 = final temperature
= final temperature
Now put all the given value in the above formula, we get:
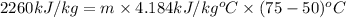
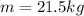
Therefore, the mass of water heated will be, 21.5 kg