Answer:
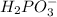 is Bronsted Lowry base.
is Bronsted Lowry base.
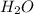 is Bronsted Lowry acid.
is Bronsted Lowry acid.
Step-by-step explanation:
According to the Bronsted Lowry conjugate acid-base theory:
- An acid is defined as a substance which donates protons and form conjugate base
- A base is defined as a substance which accepts protons and forms conjugate acid.
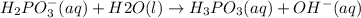
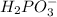 is Bronsted Lowry base.It accepts protons and forms conjugate acid
is Bronsted Lowry base.It accepts protons and forms conjugate acid
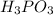
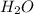 is Bronsted Lowry acid.It donates protons and forms conjugate base
is Bronsted Lowry acid.It donates protons and forms conjugate base
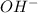

There in no exchange of proton in an above reaction.Neither of the reactants and products are Bronsted Lowry acid or Bronsted Lowry base