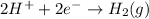Answer:
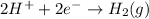
Step-by-step explanation:
Oxidation-reduction reaction or redox reaction is defined as the reaction in which oxidation and reduction reactions occur simultaneously.
Oxidation reaction is defined as the reaction in which a substance looses its electrons. The oxidation state of the substance increases.
Reduction reaction is defined as the reaction in which a substance gains electrons. The oxidation state of the substance gets reduced.
For the given reactio:
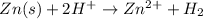
On reactant side:
Oxidation state of zinc= 0
Oxidation state of hydrogen = +21
On product side:
Oxidation state of zinc = +2
Oxidation state of hydrogen = 0
The oxidation state of hydrogen reduces from +1 to 0, it is getting reduced. Thus, it is getting reduced and it undergoes reduction reaction
Hence, the correct answer is