Answer: The moles of given hydrocarbon is 0.3 moles
Step-by-step explanation:
To calculate the number of moles, we use the equation:
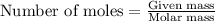
We are given:
Given mass of ethane = 10.0 g
Molar mass of ethane =
![[(2* 12)+(6* 1)]=30g/mol](https://img.qammunity.org/2020/formulas/chemistry/high-school/v93nw5ola6dnjibsha6y6yhde4b6rcysii.png)
We need to divide the given value by the molar mass.
Putting values in above equation, we get:
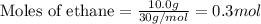
In case of multiplication and division, the number of significant digits is taken from the value which has least precise significant digits. Here, the least precise number of significant digits are 1.
Hence, the moles of given hydrocarbon is 0.3 moles