Answer: The energy required to vaporize the given amount of water is 22600 J
Step-by-step explanation:
The conversion involved in this process are:
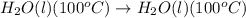
To calculate the amount of heat required at constant temperature, we use the equation:
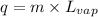
where,
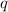 = amount of heat absorbed = ?
= amount of heat absorbed = ?
m = mass of water = 10 g
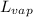 = latent heat of vaporization = 2260 J/g
= latent heat of vaporization = 2260 J/g
Putting all the values in above equation, we get:
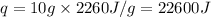
Hence, the energy required to vaporize the given amount of water is 22600 J