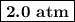Answer:
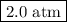
Step-by-step explanation:
If the temperature is constant, the only variables are pressure and volume, so we can use Boyle’s Law.
p₁V₁ = p₂V₂
Data:
p₁ = 4.4 atm; V₁ = 32.14 L
p₂ = ?; V₂ = 72 L
Calculation:
4.4 × 32.14 = p₂ × 72
141 = 72p₂
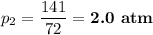
The new pressure is