Answer:

Step-by-step explanation:
To convert from moles to molecules, we must use Avogadro's Number.
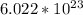
This tells us the number of particles (atoms, molecules, ions, etc) in 1 mole. In this problem, the particles are molecules of bromine in 1 mole of bromine.
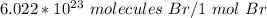
1. Convert from moles to molecules
Use Avogadro's number as a fraction or ratio.
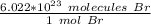
Multiply this fraction by the given number of bromine molecules.
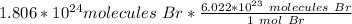
Flip the fraction so the molecules of bromine can cancel out.
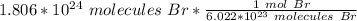
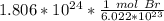
Multiply and condense the expression into 1 fraction.
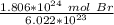
Divide.
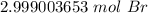
2. Round
The original measurement, 1.806*10^24 has 4 significant figures (1, 8, 0, and 6). We must round our answer to 4 sig figs. For the answer we found, that is the thousandth place.
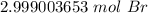
The 0 in the ten-thousandth place tels us to leave the 9 in the thousandth place.
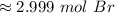
There are about 2.999 moles of bromine in 2.806 *10^23 molecules.