Answer: 32 kcal
Step-by-step explanation:
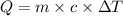
Q= heat gained or lost
m= mass of the substance = 874.3 g
c = heat capacity of water = 1 cal/g ° C
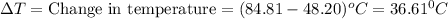
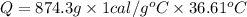
Q= 32008.1cal=32..0kcal (1kcal=100cal)
Thus heat lost by 874.3 g of iron is = 32 kcal
Heat lost is represented as a negative sign.