Assuming the temperature is kept constant during this change, you can use Boyle's Law to answer the question. The law relates the products of volume (V) and pressure (P) before and after:
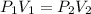
Given V1, P1, and V2:
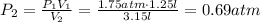
So, the pressure after the volume change, while the temperature is constant, will be 0.69 atmospheres. This is intuitive: the volume has increased with same amount of ideal gas, and so the pressure went down.