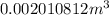Answer : The correct option is, (A)
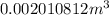
Solution : Given,
Volume of mercury at temperature
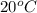 =
=
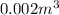
As we know that the mercury is a liquid substance. So, we have to apply the volume of expansion of the liquid.
Formula used for the volume expansion of liquid :
![V_(T)=v_(1)[1+\gamma (T_(2)-T_(1))]](https://img.qammunity.org/2020/formulas/physics/middle-school/ikt8328ee58q3w6nmzxkbdmiskpbqvzgpd.png)
or,
![V_(2)=V_(1)[1+\gamma (T_(2)-T_(1))]](https://img.qammunity.org/2020/formulas/physics/middle-school/73np1vdo468qiz386m5eo1eoml92kmmxbg.png)
where,
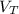 = volume of liquid at temperature
= volume of liquid at temperature
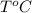
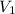 = volume of liquid at temperature
= volume of liquid at temperature
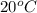
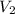 = volume of liquid at temperature
= volume of liquid at temperature
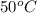
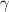 = volume expansion coefficient of mercury at
= volume expansion coefficient of mercury at
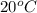 is 0.00018 per centigrade (Standard value)
is 0.00018 per centigrade (Standard value)
Now put all the given values in the above formula, we get the volume of mercury at
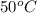 .
.
![V_(2)=0.002[1+0.00018(50-20)]=0.0020108m^3](https://img.qammunity.org/2020/formulas/physics/middle-school/wg0ok474cexld60hw4ufoiw69vpxur82ha.png)
Therefore, the volume of mercury at
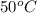 is,
is,