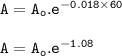The rate of decomposition of XO2 after 1 hour :
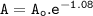
Further explanation
Given
the half-life 38.6 min
time of decomposition = 1 hour
Required
the rate of decomposition
Solution
First-order reaction :
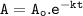
the half life=t1/2 :
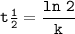
so the rate constant (k) :
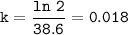
The rate after 1 hour=60 min :