Answer: 25 moles of product are formed.
Step-by-step explanation:
The balanced chemical equation will be :
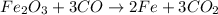
According to stoichiometry :
1 mole of
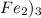 require = 3 moles of
require = 3 moles of
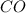
Thus 5 moles of
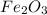 will require=
will require=
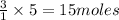 of
of
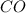
Thus
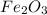 is the limiting reagent as it limits the formation of product and
is the limiting reagent as it limits the formation of product and
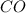 is the excess reagent.
is the excess reagent.
As 4 moles of reactant give = 5 moles of product
Thus 20 moles of reactants will give =
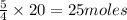 of products
of products