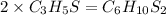Answer: The molecular formula is
Step-by-step explanation:
We are given:
Mass of
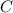 = 0.1605 g
= 0.1605 g
Mass of
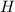 = 0.0220 g
= 0.0220 g
mass of
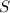 = 0.1425 g
= 0.1425 g
Step 1 : convert given masses into moles.
Moles of C =
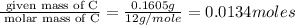
Moles of H =
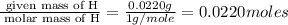
Moles of S =
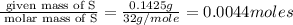
Step 2 : For the mole ratio, divide each value of moles by the smallest number of moles calculated.
For C =
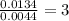
For H =
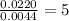
For S =
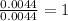
The ratio of C : H: S= 3: 5: 1
Hence the empirical formula is
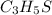
The empirical weight of
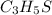 = 3(12)+5(1)+1(32)= 73g.
= 3(12)+5(1)+1(32)= 73g.
The molecular weight = 146 g/mole
Now we have to calculate the molecular formula.
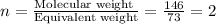
The molecular formula will be=