Answer:
1.05 mols of O2 gas
Step-by-step explanation:
For this type of problem, it's important to understand what the balanced Chemical equation tells us:
Balanced Chemical equations
Let's look at the balanced chemical equation:
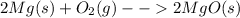
This equation two sides of the reaction arrow. On the left are the reactants (things you start with, and that react during the chemical reaction), and on the right side are products (things that are produced during the chemical reaction that you end with).
The numbers in front of each compound tell how many of molecules are involved for a full reaction without anything left over. A "mol" is a large quantity (
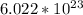 ), in this case, of molecules , since it's unlikely you're only taking a single molecule of each substance (it would be so tiny, you wouldn't even know you were doing the reaction).
), in this case, of molecules , since it's unlikely you're only taking a single molecule of each substance (it would be so tiny, you wouldn't even know you were doing the reaction).
So,for every 2 moles of Magnesium used, we'll also need 1 mole of Oxygen, and it will produce 2 moles of Magnesium Oxide.
In a way, during the reaction it's almost like 2 moles of Magnesium is equal to 1 mole of Oxygen and is equal to 2 moles of Magnesium Oxide:
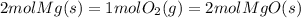
From here, we can build some unit ratios, to convert between the known quantity of moles we have, and find the unknown quantity of moles that are requested.
Finding the right unit ratio
We know that we are looking at 2.10 mol of Magnesium, so we want a unit ratio with moles of Mg on the bottom. We want to find moles of O2, so we want a unit ratio of moles of O2 on top.
The unit ratio we want is the middle part of the equation, divided by the left part of the equation.
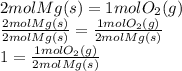
Since this quantity is 1, it is a unit ratio and can be multiplied to other things to change their units (for this problem).
Finding the answer
Starting with what we know, and multiplying by our unit ratio:
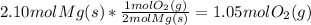
Notice that the units from the first quantity cancel with the units on the bottom of the fraction, leaving only the unit on top of the fraction ... the exact units we wanted!
So, 1.05mols of O2 would be consumed during the reaction if exactly 2.10moles of Magnesium are burned.