Answer:
9.6 mol
Step-by-step explanation:
Start by writing out the balanced equation of the reaction.
2
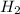 +
+
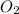 ⇒ 2
⇒ 2
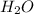
Here you will see 2 mol of hydrogen for every 1 mol of oxygen. Using this ratio, you can assume the amount of hydrogen will always be double that of oxygen. In this case, 4.8 * 2 = 9.6