The translation of the question is
250 g of alcohol have been put in a glass together with 2 g of iodine, which dissolve completely. Calculate the concentration of the solution in% by mass?
Answer:
The %mass by mass of the given solution is 0.794%
Step-by-step explanation:
We are given
Mass of solute(Iodine) = 2g
Mass of the solvent (Alcohol) = 250g
Since the Iodine dissolves completely in Alcohol
Total mass of the solution = 250 + 2 = 252g
the formula of concentration of the solution in % by Mass is given by
%
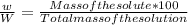
%w/W =
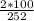
= 0.794%
Therefore the %mass by mass of the given solution is 0.794%