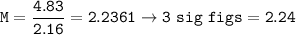The molarity of a solution : 2.24
Further explanation
Molarity shows the number of moles of solute in every 1 liter of solution or mmol in each ml of solution
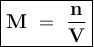
Where
M = Molarity
n = number of moles of solute
V = Volume of solution (L)
4.83 moles of NaCl in 2.16 L solution
n = 4.83
V = 2.16 L
Molarity :