Answer:
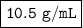
Step-by-step explanation:
Density can be found by dividing the mass by the volume.
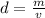
The mass is 42 grams.
Let's find the volume. The volume was found through water displacement, so we must subtract the final volume of the water with the bracelet (14 mL) and the starting volume of water (10 mL)
Now we know that:
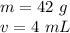
Substitute the values into the formula.
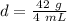
Divide.
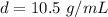
The density of the silver is 10.5 grams per milliliter.