Answer:
(1). The pressure is 533 KPa.
(2). The temperature is 406 K.
Step-by-step explanation:
Given that,
Number of moles = 4.52 moles
Volume = 21.0 L
Temperature = 298 K
(1). We need to calculate the pressure
Using ideal equation of gas
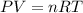
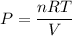
Where, T = temperature
R = gas constant
V = volume
n = number of moles
Put the value into the formula
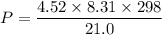
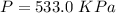
(2). Number of moles = 1.60 moles
Volume = 21.0 L
Temperature = 298 K
We need to calculate the temperature
Using ideal equation of gas
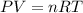
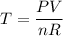
Put the value into the formula
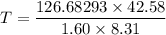
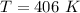
Hence, (1). The pressure is 533 KPa.
(2). The temperature is 406 K.