Answer:
c = 14.668 J/kg°C
Step-by-step explanation:
Given that,
Heat added, Q = 965 J
Mass of the sample, m = 0.25 kg
The change in temperature,
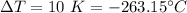
We need to find the specific heat of the copper. The heat required to raise the temperature is given by :
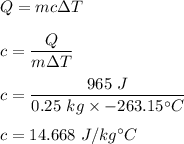
So, the specific heat of the copper is 14.668 J/kg°C.