Answer:
6.2 g
Step-by-step explanation:
The mass of oxygen gas produced from 8.5 g CO2 would be 6.2 g.
First, let us look at the equation of the reaction:
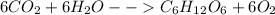
From the equation, 6 moles of CO2 are required to produce 6 moles of O2.
Recall that: mole = mass/molar mass
8.5 g of CO2 = 8.5/44.01 = 0.1931 mole of CO2
Since the ratio of CO2 to O2 is 1:1, 0.1931 moles of CO2 will produce 0.1931 moles of O2.
Hence, mass of oxygen produced = mole x molar mass
= 0.1931 x 32
= 6.2 g to 2 significant digits.