Answer:
2.7
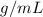
Explanation:
The density of an object will be its mass over volume.
We know that the mass of this rock is 54 grams because it took 54 grams of gram stackers to balance the scale. If we have a balanced scale, that means the amount of weight on either side is equal. So the mass is 54 grams.
Also, we can tell that the volume of the rock is 20 mL. This is because, according to Archimedes principle, if you put an object in water, the amount of water displacement will be it's volume. So the amount of water displacement is
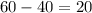 mL.
mL.
Density is mass over volume, so the density is
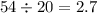 . Density is measure in mass over volume, so
. Density is measure in mass over volume, so
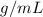 .
.
So 2.7
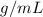 is the density of the rock.
is the density of the rock.
Hope this helped!