Answer:
The volume required for complete neutralize is 32.29 mL
Step-by-step explanation:
The computation of the volume required for complete neutralize is shown below:
As we know that, the balanced equation is
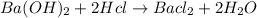
Now
The number of moles of
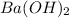 = n_1 = 1
= n_1 = 1
And, the number of moles of Hcl = n_2 = 2
Therefore
The equation i.e. to be used to find out the volume is given below:
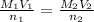
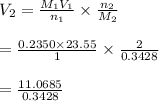
= 32.29 mL
Hence, the volume is 32.29mL