Answer : The correct option is, (c) 1
Explanation :
Isotope : It is defined as the element that have the same number of protons but have the different number of neutrons of each of the atom.
Isotope notation : It is also called as nuclear notation. In isotopic notation we can easily determine an isotope's mass number, atomic number, the number of neutrons and protons in the nucleus.
The general isotopic representation of an atom is:
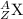
where,
Z = Atomic number of the atom
A = Mass number of the atom
X = Symbol of the atom
As we are given two isotopes:
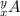 and
and
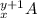
From this we conclude that,
The number of neutrons in isotope
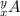 = y-x
= y-x
The number of neutrons in isotope
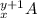 = (y+1)-x
= (y+1)-x
Difference between the number of neutrons in the isotopes = [(y+1)-x] - [y-x] = 1
Therefore, the difference between the number of neutrons in the isotopes is 1.