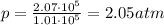Answer:
2.05 atm
Step-by-step explanation:
The pressure exerted by a force is equal to the rate between the force exerted and the area over which the force is exerted:
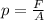
where
p is the pressure
F is the force
A is the area
In this problem, we have the pressure written as
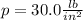
First, we need to convert this into SI units (Newton over squared meters, which is Pascal).
We have:
1 lb = 4.45 N
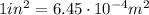
So the pressure converted into SI units is
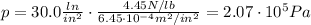
Now we know that 1 atmosphere is equivalent to
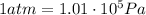
So we can convert this pressure into atmospheres: