Answer: The new pressure when temperature remains constant is 3.56 atm
Step-by-step explanation:
To calculate the new pressure, we use the equation given by Boyle's law. This law states that pressure is inversely proportional to the volume of the gas at constant temperature.
The equation given by this law is:
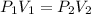
where,
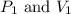 are initial pressure and volume.
are initial pressure and volume.
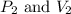 are final pressure and volume.
are final pressure and volume.
We are given:
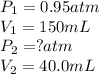
Putting values in above equation, we get:
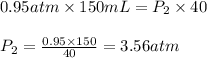
Hence, the new pressure when temperature remains constant is 3.56 atm