Answer :
The coefficients are, 1 in Ca, 2 in HF, 1 in CaF₂ and 1 in H₂
The subscripts are, 2 in CaF₂ and 2 in H₂
The states of reactants are, Ca in solid state (s) and HF in aqueous state (aq).
The evidence that a reaction occurred is, the new substances are formed in the chemical reaction.
Explanation :
Balanced chemical reaction : It is defined as the reaction in which the number of atoms of individual elements present on reactant side must be equal to the product side.
If the amount of atoms of each type on the left and right sides of a reaction differs then to balance the equation by adding coefficient in the front of the elements or molecule or compound in the chemical equation.
The coefficient tell us about that how many molecules or atoms present in the chemical equation.
Coefficient : It is a number that written in front of the element, molecule or ion.
Subscript : It is a small number that placed slightly lower-right than the normal text.
The given balanced chemical reaction is:
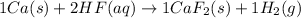
In this reaction:
The coefficients are, 1 in Ca, 2 in HF, 1 in CaF₂ and 1 in H₂
The subscripts are, 2 in CaF₂ and 2 in H₂
The states of reactants are, Ca in solid state (s) and HF in aqueous state (aq).
The evidence that a reaction occurred is, the new substances are formed in the chemical reaction.