Answer : The millimoles of acetate need to add to this solution will be, 18.2 mmol
Explanation :
For acetic acid,
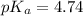
Moles of of acetic acid = 10 mmol
pH = 5.00
Using Henderson Hesselbach equation :
![pH=pK_a+\log ([Salt])/([Acid])](https://img.qammunity.org/2021/formulas/biology/college/z944fnahhldpjolfrvealc6q9baj5h69q3.png)
Now put all the given values in this expression, we get:
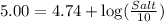
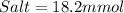
Thus, the millimoles of acetate need to add to this solution will be, 18.2 mmol