Answer : The final concentration of
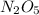 is, 2.9 M
is, 2.9 M
Explanation :
Expression for rate law for first order kinetics is given by:
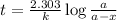
where,
k = rate constant =
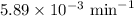
t = time passed by the sample = 3.5 min
a = initial concentration of the reactant = 3.0 M
a - x = concentration left after decay process = ?
Now put all the given values in above equation, we get
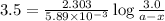
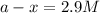
Thus, the final concentration of
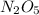 is, 2.9 M
is, 2.9 M