Answer:
Step-by-step explanation:
The atomic mass of nickel is 58.963g/mol. Thus, one mole of nickel weighs "exactly" 58.693g.
How much nickel does every coin contain?
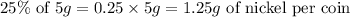
How many times 58.693g contains 1.25g?
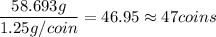
That means that you need to have 47 coins of nickel to have one mole of nickel.