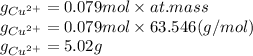Answer:
The amount of Cu²⁺ plated out is 5.02 g.
Step-by-step explanation:
To solve the problem we must first consider the following equation
Cu²⁺ + 2e -------> Cu (s) ---------- (1)
This equation shows that two moles of electrons are used for the conversion of Cu²⁺. So let us first determine the number of moles of electrons, which can be calculated from the following formula
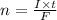
Here, I is the current in ampere, t is time in seconds and F is the Faraday's constant. Placing the data we get,
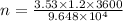
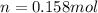
Equation 1 shows that 1 mole of Cu²⁺ consumes two moles of electrons. So, the moles of Cu²⁺ will be half of the moles of electrons.
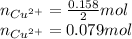
the mass of Cu²⁺ will be