Step-by-step explanation:
Chemical formula of a permanganate ion is
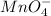 and this ion has an overall charge of -1. On the other hand, calcium is an alkaline earth metal and it has a valency of +2. Therefore, when it will chemically combines with permanganate ion then according to cross-multiplication formula it will be multiplied by 2.
and this ion has an overall charge of -1. On the other hand, calcium is an alkaline earth metal and it has a valency of +2. Therefore, when it will chemically combines with permanganate ion then according to cross-multiplication formula it will be multiplied by 2.
Therefore, we can conclude that the formula of the ionic compound containing calcium and permanganate ions is
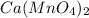 .
.