Step-by-step explanation:
For a compound to show hydrogen bonding it is necessary that the hydrogen atom of the compound should be attached to more electronegative atom like fluorine, oxygen or nitrogen.
For example,
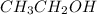 ,
,
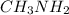 and
and
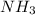 all these compounds contain an electronegative atom attached to hydrogen atom.
all these compounds contain an electronegative atom attached to hydrogen atom.
Therefore, these pure compounds will exhibit hydrogen bonding.
Thus, we can conclude that out of the given options
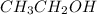 ,
,
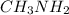 and
and
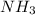 are the pure compounds which will exhibit hydrogen bonding.
are the pure compounds which will exhibit hydrogen bonding.