Answer:
2726.54 J/K
Step-by-step explanation:
Entropy change =
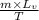
where
m = mass of water = 450g = 0.45kg
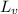 = 2.26 MJ/kg = 2260000 J/kg
= 2.26 MJ/kg = 2260000 J/kg
T = temperature at which water change from liquid to steam = 100 degree Celsius = 272 + 100 = 373 degree kelvin.
Entropy change =
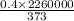 = 2726.54 J/K
= 2726.54 J/K