Answer:
Option A.
Step-by-step explanation:
In quantum physics there is a law to relate the position and the momentum of the particle, it says that if we know with precision where is a quantum particle, we can not know the momentum of this particle, in other words, the velocity of the particle. So, when we measure the velocity of the particle we find the correct value of the particle, but we can not determine with accuracy where is the particle. This law is known as the Heisenberg's uncertainty principle and, its expressed as follows:
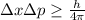
where Δx: is the position's uncertainty, Δp: is the momentum's uncertainty and h: is the Planck constant.
Therefore, the correct answer is A: measuring the velocity of a tiny particle with an electromagnet has no effect on the velocity of the particle. It only affects the determination of the particle's position.
I hope it helps you!