Answer:
V = 0.24 L.
Step-by-step explanation:
Hello there!
In this case, it is possible to approach this problem by using the ideal gas equation defined by:
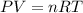
Whereas our unknown is V, volume, and we solve for it as follows:
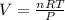
Then since the STP conditions are ate 273.15 K and 1 atm, the volume of 0.011 moles of CO2 gas turns out to be:
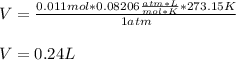
Regards!