Answer:
r = 0.5297 m
Step-by-step explanation:
In this exercise we use Newton's second law where the force is magnetic
F = ma
centripetal acceleration
a = v² / r
F = q v x B = q v B sin θ
where the angle between the velocity and the magnetic field is 90º, therefore the sin 90 = 1
we substitute
q v B= m v² / r
r =
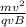
the mass of each isotope is
12C
m12 = 6 m_proton + 6 m_neutrons
m12 = (6 1,673 +6 1,675) 10⁻²⁷
m12 = 20.088 10-27 kg
14C
m14 = 6 m_proton + 8 m_neutron
m14 = (6 1,673 + 8 1,675) 10-27
m14 = 23,438 10⁻²⁷ kg
in the exercise they indicate that the velocity of the two particles is the same, therefore with the initial data we can calculate the parameters that do not change in the experiment.
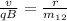
v / qB = 0.454 / 20.088 10⁻²⁷
v / qb = 2.26 10²⁵
this quantity remains constant, let's use the other data to calculate the radius
r = 23.438 10⁻²⁷ 2.26 10²⁵
r = 5.297 10⁻¹ m
r = 0.5297 m